|
Furthermore, a well-accepted correlation exists between the 3D upregulation of cell–cell and cell–matrix interactions and increased MSC paracrine benefit compared to 2D monolayer culture. Tissue culture-treated 2D culture surfaces impose high basal adhesion and forced cell polarity 3D culture systems diminish forced polarity and excessive basal spreading, allowing 3D cells to adopt a more physiologic morphology favorable for MSC paracrine function 17, 18. Importantly, these systems have also been shown to enhance MSC paracrine activity through both morphological and molecular changes related to cell interactions compared to conventional two-dimensional (2D) adherent culture 14, 15, 16. MSC cultivation with three-dimensional (3D) culture systems, predominantly scaffolds, hydrogels, and spheroids, is familiar for tissue engineering applications that overcome engraftment limitations of cell injections. Without high transplantation efficacy of potent, clinically meaningful cell numbers, continuous paracrine factor delivery is not possible, thereby blunting the potential efficacy of MSC therapies 13. Despite some clinical benefits, MSC engraftment following direct injection is generally low and transient in nature 10, 11, 12. For example, clinical trials to treat ischemic heart failure have demonstrated that MSC injection into the border zone between infarcted and viable cardiac tissue resulted in potent antifibrotic effects, including augmentation of viable and perfused tissue, despite no injected MSC engraftment or differentiation cardiac improvement is instead attributed to endogenous regeneration mechanisms stimulated by MSC-secreted bioactive factors 9. A growing body of clinical evidence attributes the therapeutic role of MSCs to their paracrine actions 6, 7, 8. A unique characteristic of MSCs is their ability to secrete a wide range of bioactive molecules, including growth factors and cytokines, that can influence nearby cells via paracrine signaling to facilitate various biological processes desirable for tissue regeneration, such as angiogenesis, immune modulation, injured cell repair rather than death and scarring, cell proliferation, and cell recruitment and differentiation 2, 3, 4, 5. With the expansion of tissue engineering and regenerative medicine, mesenchymal stem cells (MSCs) have gained traction as effective candidates for repair and regeneration of injured tissues 1. Together, these findings demonstrate that MSC therapeutic potency can be enhanced by 3D cell sheet tissue structure. 3D culture increased MSC gene expression of cell interaction proteins, β-catenin, integrin β1, and connexin 43, and of pro-tissue regenerative cytokines, vascular endothelial growth factor (VEGF), hepatocyte growth factor (HGF), and interleukin-10 (IL-10), and increased VEGF secretion per MSC 2.1-fold relative to 2D cultures. This 2D-to-3D transition reorganized MSC actin cytoskeleton from aligned to multidirectional, corresponding to a cell morphological change from elongated in 2D monolayers to rounded in 3D cell sheets. Release of culture-adherent tension induced spontaneous cell sheet contraction, reducing the diameter 2.4-fold, and increasing the thickness 8.0-fold to render a 3D tissue-like construct with a 36% increase in tissue volume. MSCs cultivated on temperature-responsive culture dishes to a confluent 2D monolayer were harvested by temperature reduction from 37 to 20 ☌ that induces a surface wettability transition from hydrophobic to hydrophilic. 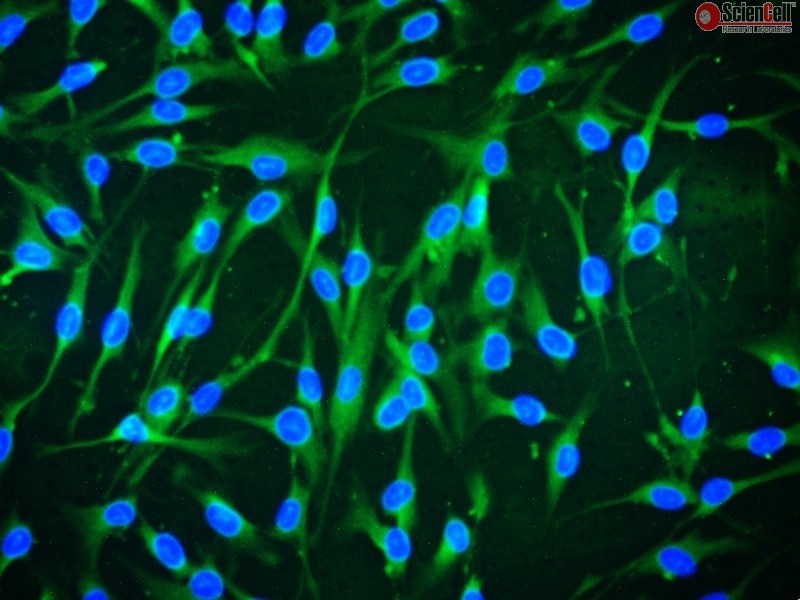
Toward this end, this study explored scaffold-free cell sheet technology as a new 3D platform. 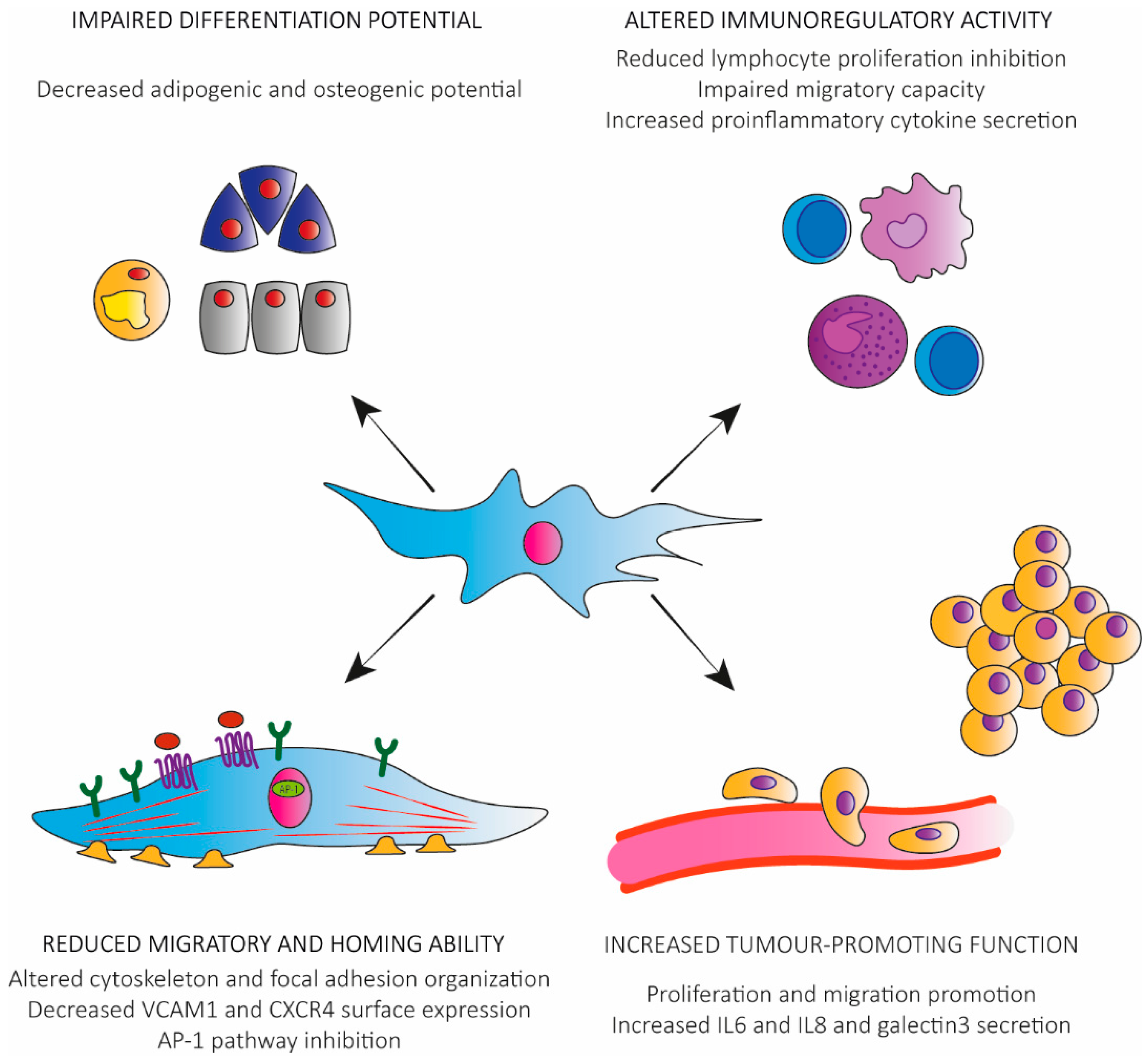
An increasing body of evidence suggests that this paracrine function is enhanced by MSC cultivation in three-dimensional (3D) tissue-like microenvironments. Mesenchymal stem cells (MSCs) secrete paracrine factors that play crucial roles during tissue regeneration.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
